The expansion of telemedicine use in NMDs and development of clinically relevant but easy-to-use remote monitoring systems has potential to improve patient access to expert care.
M. S. Damian, MD, FEAN, FNCS, and Prof. P. Laforet, MD, PhD
Patients with neuromuscular disorders (NMDs) have diverse and complex care requirements, typically served by highly specialized centers. However, these may be geographically remote, and the COVID-19 pandemic underlined the system’s fragility by leading to widespread suspension of diagnostic, support, and rehabilitative services.1 Cancelled routine visits and limited outreach resulted in morbidity and even deaths. The demand for telehealth and remote care to help resolve this situation increased. This article describes the concept and current options using telemedicine in the care of people with NMDs.
Telemedicine in NMD at the Onset of the COVID-19 Pandemic
Prior to COVID-19, telemedicine was mainly used to overcome geographical challenges in thinly populated or resource-poor regions, or for monitoring patients with impaired mobility at home. Small case series suggested efficacy and economic advantages by reducing hospitalization. Zamarron et al. demonstrated the long-term feasibility of telemonitoring with video consultations plus Sp02, BP, ECG, overnight oximetry in home-ventilated patients via a residential internet gateway with alarm system and monthly outreach nurse check-ups.2 Challenges included changes in the patient-carer relationship, and difficulty procuring individually adapted systems.
COVID-19 forced a hasty rethink of this position. Guidon and Amato discussed neuromuscular telemedicine consultation by phone or videolink.3 They perceived high potential for follow-up in myopathy, myasthenia gravis (MG), and polyneuropathy when stable, or for management of pain; moderate utility for new or unstable neuromuscular disorders, but low utility where there were discrepant symptom findings, for second opinions, and for primary management of unstable patients. Face-to-face clinics remained mandatory where electrodiagnostic studies and muscle and nerve biopsies were urgently needed, and where the results would change management, as in new onset amyotrophic lateral sclerosis (ALS), MG, and immune-mediated neuropathy or myopathy.
New scores and protocols may mitigate these shortcomings: Garibaldi et al. developed functional scores for myopathies and neuropathies (the Myo-FRS and N-FRS), taking reference to older disease-specific scores such as the MG-ADL for myasthenia gravis, and the ALSFR-R for amyotrophic lateral sclerosis.4 Ricciardi et al. suggested a protocol for remote clinical testing in MG, featuring:
- Counting aloud test in one breath (CAT)
- Hoarseness test (voice change with high-pitched vocalization)
- Head-up test (10s head flexion from supine)
- Swallowing test (3oz = 90ml water swallow)5
Other approaches included the Veteran Affairs Neuropathy Scale, which Wilson et al. piloted in telemedicine clinics6 and teleswallowing, a remote swallow assessment.7 This work provides a toolkit to perform a detailed clinical assessment, remotely via videolink. Purely audio remote interviews are more limited. Significant technical challenges for remote clinics remain regarding the availability of monitoring devices, broadband speed, audio-visual quality, internet lagtime for timed tests (10m walk, Timed-up and Go-test), users’ technical expertise, and computer literacy. To improve this, protocols to perform a video NMD clinic have been published.8,9 Videoconferencing platforms have been evaluated,10,11,12 and video platforms are available in a number of commercial patient management systems. Overall, the use of telemedicine in NMDs increased during the COVID-19 pandemic, but telemonitoring was used considerably less.13
Telemonitoring found its first application in clinical trials to optimize remote clinical assessment, but also to improve trial recruitment and monitoring. Reliability and strong correlations between wearable physical activity monitors (sensor-based systems using activity watches or body-worn sensors, PAMs) and neuromuscular measures confirmed PAMs’ utility as outcome measures and in long term monitoring.14 Mobility data can be gathered by PAMs, or by ambient measurement systems (AMS), which passively measures movement such as ambulation speed, rise-to-stand speed, and arm-raise speed when someone is in range of a sensor.
Remote monitoring of life-supporting technology, such as home mechanical ventilation (HMV), requires regular monitoring of physiological variables (spO2, spCO2, respiratory rate) by carers supported by specialist outreach, and requires a continuous data link to the monitoring center for analysis and troubleshooting.15 It can enable remote initiation of HMV and may reduce costs, and may help predict exacerbations, allow remote interventions and adjustments.16,17 Challenges about data security and privacy, caregiver involvement and acceptance, availability of high-speed internet, and misconceptions around time needed, remain.18,19
Mobile Phone-Based Clinical Assessment
Wearable monitors have the disadvantage that they are expensive pieces of advanced technology, and the proliferation of devices patients must wear to allow multimodal monitoring can be intrusive. This makes them both cumbersome as well as unsuitable for low-income health environments. Therefore, exploring the potential of a ubiquitous device, such as a smartphone to provide multimodal monitoring is attractive.
Digital technologies are currently expanding rapidly, especially in the field of NMDs. They can reduce data collection burden and increase knowledge of real-life data. MG is an autoimmune neuromuscular disease characterized by very heterogeneous symptoms potentially associating ocular, bulbar, respiratory and skeletal muscles weakness and fatigability.
In current practice, visits to the physician’s office are planned every three to six months. However, since patients might experience worsening symptoms outside of visits, clinicians must often rely on patient recollection during consultations, which present a recall and subjectivity bias that can compromise the estimation of disease status. In this context, it will be clinically relevant to allow patients to self-assess their symptoms and physicians to collect and analyze digital biomarkers for a closer monitoring.
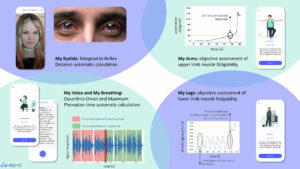
Figure 1. Remote clinical assessment enabled on a smartphone.
As an example, an ongoing study (ME&MG™, NCT: 05564936) aims to validate a digital solution that runs on patients’ smartphones. It is intended to be used as an unsupervised digital self-assessment tool for the monitoring of muscle weakness, fatigability, and disability in patients living with MG. This application contains digital active tests for the assessment of ptosis, breathing, dysarthria, upper- and lower-limb weakness, treatment follow-up, and validated e-questionnaires related to daily activities, pain, fatigue, sleep, and depression. The objectives of this study are to validate the clinical performance of the unsupervised at-home self-assessment of symptoms on the patient’s smartphone with ME&MG™ compared to the standard in-clinic testing, including analytical performance as well as to evaluate the safety of the solution, its usability, and satisfaction. Eight sites in France and the United States will be involved in this study.20 A further study evaluating the device is ongoing in the U.S. and Canada (NCT05566964).21
Figure 1 demonstrates the remote clinical assessment enabled on smartphone using the ME&MG software.
Machine-Learning Models of Telemonitoring and AI-Based Analysis of Digital Biomarkers
Machine-learning (ML) and AI-based models can conceivably help establish objective, rapid, and more accurate interpretation of remote data acquired by telemedicine monitoring. Vieira et al. devised an objective measure for ALS disease severity based on voice samples and accelerometer measurements, correlated with ALS-FRS-R scores over a four-year period with an audio voice recording and Actigraph GT3X accelerometers on each limb. They also trained ML models to predict bulbar-related and limb-related ALSFRS-R scores.22
Similar approaches were used to assess changes in an edaravone-treated patient sample. Wearables can produce an objective severity score.23 There have been several approaches to wearables for therapy studies,24 though robust validation is still awaited.
Conclusion
The expansion of telemedicine use in NMDs and development of clinically relevant but easy-to-use remote monitoring systems has potential to improve patient access to expert care, even in situations where direct face-to-face access is interrupted, as in the recent pandemic, or where scarce resources or geography prevents patient access to specialist care. Going forward, telemedicine might expand the availability of high-quality specialist care to patients in low-income societies, who hitherto have had little access. Recent developments in effective treatment can also be seen as an obligation on global medicine to explore how inequality in provision can be mitigated — telemedicine technology may advance us one step in this direction. •
M. S. Damian, MD, FEAN, FNCS, works at the Essex Cardiothoracic Centre, in Basildon, UK, and Prof. P. Laforet, MD, PhD, works in the Neurology Department, Raymond Poincaré Hospital, APHP, Garches, APHP, FHU PHENIX, Centre de référence des maladies neuromusculaires Nord Est Ile-de-France in Filnemus.
References
- Mauri E, Abati E, Musumeci O, et al. Estimating the impact of COVID-19 pandemic on services provided by Italian Neuromuscular Centers: an Italian Association of Myology survey of the acute phase. Acta Myol. 2020 Jun 1;39(2):57-66. doi: 10.36185/2532-1900-008.
- Zamarrón C, Morete E, González F. Telemedicine system for the care of patients with neuromuscular disease and chronic respiratory failure. Arch Med Sci. 2014 Oct 27;10(5):1047-51. doi: 10.5114/aoms.2014.46223.
- Guidon AC, Amato AA. COVID-19 and neuromuscular disorders. Neurology. 2020 Jun 2;94(22):959-969. doi: 10.1212/WNL.0000000000009566.
- Garibaldi M, Siciliano G, Antonini G. Telemedicine for neuromuscular disorders during the COVID-19 outbreak. J Neurol. 2021 Jan;268(1):1-4. doi: 10.1007/s00415-020-10063-7.
- Ricciardi D, Casagrande S, Iodice F, et al. Myasthenia gravis and telemedicine: a lesson from COVID-19 pandemic. Neurol Sci. 2021 Dec;42(12):4889-4892. doi: 10.1007/s10072-021-05566-8.
- Wilson AM, Ong MK, Saliba D, Jamal NI. The Veterans Affairs Neuropathy Scale: A Reliable, Remote Polyneuropathy Exam. Front Neurol. 2019 Nov 1;10:1050. doi: 10.3389/fneur.2019.01050.
- www.teleswallowing.com
- Damian MS. Neuromuscular monitoring devices – where to go next? Curr Opin Neurol. 2021 Oct 1;34(5):721-726. doi: 10.1097/WCO.0000000000000977.
- Al Hussona M, Maher M, Chan D, et al. The Virtual Neurologic Exam: Instructional Videos and Guidance for the COVID-19 Era. Can J Neurol Sci. 2020 Sep;47(5):598-603. doi: 10.1017/cjn.2020.96.
- Fortunato F, Bianchi F, Ricci G, et al. Digital health and Clinical Patient Management System (CPMS) platform utility for data sharing of neuromuscular patients: the Italian EURO-NMD experience. Orphanet J Rare Dis. 2023 Jul 21;18(1):196. doi: 10.1186/s13023-023-02776-5.
- Reyaz A, Agarwal A, Padma Srivastava MV, et al. Impact of Tele-Neuromuscular Clinic on the Accessibility of Care for Patients with Inherited Neuromuscular Disorders during COVID-19 Pandemic in India. Ann Indian Acad Neurol. 2022 May-Jun;25(3):505-507. doi: 10.4103/aian.aian_565_21.
- Amin R, Pizzuti R, Buchanan F, Rose L. A virtual care innovation for home mechanical ventilation. CMAJ. 2021 Apr 26;193(17):E607-E611. doi: 10.1503/cmaj.202584.
- El-Hassar L, Amara A, Sanson B, et al. Telemedicine in Neuromuscular Diseases During Covid-19 Pandemic: ERN-NMD European Survey. J Neuromuscul Dis. 2023;10(2):173-184. doi: 10.3233/JND-221525.
- Rockette-Wagner B, Aggarwal R. A Review of The Evidence for the Utility of Physical Activity Monitor Use in Patients with Idiopathic Inflammatory Myopathies. Rheumatology (Oxford). 2024 Jan 18:keae004. doi: 10.1093/rheumatology/keae004.
- Angelucci A, Aliverti A. Telemonitoring systems for respiratory patients: technological aspects. Pulmonology. 2020 Jul-Aug;26(4):221-232
- Hazenberg A, Kerstjens HA, Prins SC, et al. Initiation of home mechanical ventilation at home: a randomized controlled trial of efficacy, feasibility and costs. Respir Med. 2014 Sep;108(9):1387-95. doi: 10.1016/j.rmed.2014.07.008.
- van den Biggelaar RJM, Hazenberg A, Cobben NAM, Gaytant MA, Vermeulen KM, Wijkstra PJ. A Randomized Trial of Initiation of Chronic Noninvasive Mechanical Ventilation at Home vs In-Hospital in Patients With Neuromuscular Disease and Thoracic Cage Disorder: The Dutch Homerun Trial. Chest. 2020 Dec;158(6):2493-2501. doi: 10.1016/j.chest.2020.07.007.
- van den Biggelaar R, Hazenberg A, Duiverman ML. The role of telemonitoring in patients on home mechanical ventilation. Eur Respir Rev. 2023 Apr 5;32(168):220207. doi: 10.1183/16000617.0207-2022.
- Ambrosino N, Vitacca M, Dreher M, et al. Tele-monitoring of ventilator-dependent patients: a European Respiratory Society Statement. Eur Respir J. 2016 Sep;48(3):648-63. doi: 10.1183/13993003.01721-2015.
- The ME&MG Digital Solution for Autonomous Assessment of Myasthenia Gravis. Online: https://clinicaltrials.gov/study/NCT05564936
- Descriptive Analysis of Real-world Data Collected With the ME&MG open Mobile Application Developed for Myasthenia Gravis Patients. Online: https://clinicaltrials.gov/study/NCT05566964
- Vieira FG, Venugopalan S, Premasiri AS, et al. A machine-learning based objective measure for ALS disease severity. NPJ Digit Med. 2022 Apr 8;5(1):45. doi: 10.1038/s41746-022-00588-8.
- Gupta AS, Patel S, Premasiri A, Vieira F. At-home wearables and machine learning sensitively capture disease progression in amyotrophic lateral sclerosis. Nat Commun. 2023 Aug 21;14(1):5080. doi: 10.1038/s41467-023-40917-3.
- Poleur M, Markati T, Servais L. The use of digital outcome measures in clinical trials in rare neurological diseases: a systematic literature review. Orphanet J Rare Dis. 2023 Aug 2;18(1):224. doi: 10.1186/s13023-023-02813-3.
